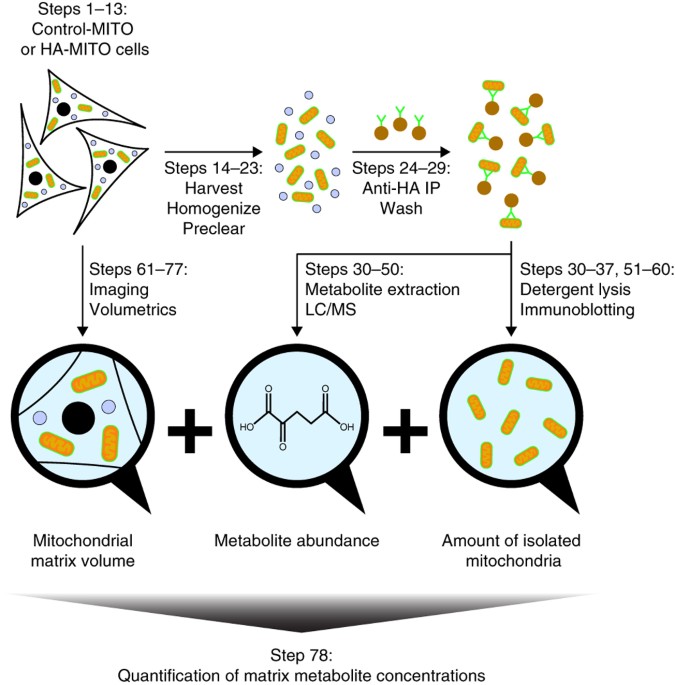
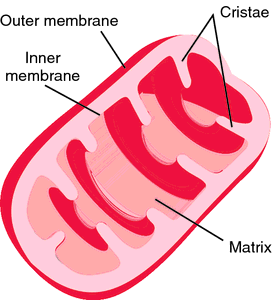
Because these features are highly conserved, this mechanism is likely to apply to the whole mitochondrial carrier family. Uniquely, the carrier switches between states by rotation of its three domains about a fulcrum provided by the substrate-binding site. Glycine and small amino acid residues allow close-packing of helices on the matrix side. The cytoplasmic side of the carrier is closed by conserved hydrophobic residues, and a salt bridge network, braced by tyrosines. Other metabolic functions of C14orf159, especially on mitochondrial metabolism, and its contribution to CRC. Here, we describe the structure of the matrix-open state locked by bongkrekic acid bound in the ADP/ATP-binding site at the bottom of the central cavity. C14orf159 is a mitochondrial matrix protein converting d-glutamate to 5-oxo-d-proline. The structure of the cytoplasmic-open state is known, but it has proved difficult to understand the transport mechanism in the absence of a structure in the matrix-open state. Mitochondrial matrix calcium levels can reach the tens of micromolar levels, which is necessary for the activation of isocitrate dehydrogenase, one of the key regulatory enzymes of the Krebs cycle. Mitochondrial ADP/ATP carriers transport ADP into the mitochondrial matrix for ATP synthesis, and ATP out to fuel the cell, by cycling between cytoplasmic-open and matrix-open states. In neurons, concomitant increases in cytosolic and mitochondrial calcium act to synchronize neuronal activity with mitochondrial energy metabolism.
